Cas No: 7738-94-5
EINESC No: 231-801-5
Molecular weight: 118,01 g/mol
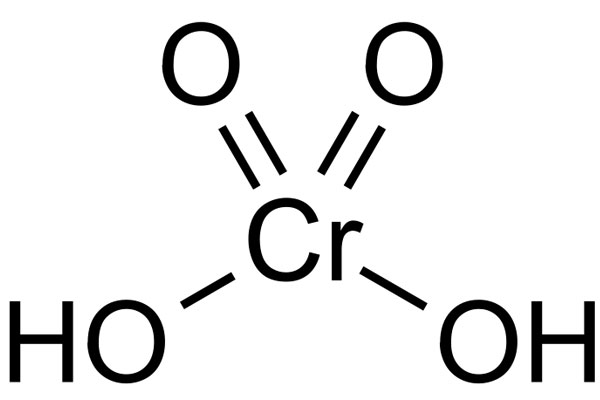
Chemical Formula: H2CrO4
Chemical Structure:
Physical Properties
General Properties: Dark red crystals
Odor: Odorless
Intensity: 1.201 g/mL3
Boiling point: 250 °C
Melting point: 197 °C
Flash point:-
Vapor pressure:-
Refraction index:-
Solubility : 169 g/100 ml
General Properties
Chromic acid generally refers to a mixture produced by adding concentrated sulphuric acid to a dichromate. Dichromate may contain several other compounds such as solid chromium trioxide. It is a very good chemical for glass cleaning. Anhydrous form of trioxide(H2CrO4) can also be called chromic acid. It is a strong and abrasive oxidizing agent. Chemically, it bear may remeblance to sulphuric acid and acts simlarly when yielding hydrogen. Only sulphuric acid yields first proton much easier than chromic acid.
Additionally, chromic acid slowly disintigrates while reaching boiling point and, in proper environments, it becomes dessicant.
Production
Chromic acid is acquired from adding additive chemicals to chrome trioxide’s aquenous solution. Chrome trioxde is generally produced by putting 2,4 mol sodium dichromate and 2,8 mol sulphuric acid.
Applications
In chemistry trade, chromic acid is used in chromate, which is salt of chromic acid, production. A large portion of chromic acid’s production is used for chrome coating. It is used as burner in medical fields due to it being a good oxidizing agent. It is also efficient in cleaning organic filth from glasses in labs but this method is not preferred because of its harm to environment. It is also used as rubber pigment in carving processes, salt glaze making, colorizing glasses, cleaning metals, ink and dye productions.
Safety Measures and Toxic Levels
Chromic acid, like all hexavalent chromium compounds, is extremely toxic and carcinogenic. Therefore, its usage is not advised except for some private sectors. It may be fatal if swallowed or inhaled. Also, when inhaled, it may cause permanent lung diseases such as asthma. In eye contact, it may cause permanent blindness. In skin contact, it may cause serious injuries or allergic reactions. It also may cause cancer and greatly reduce fertility. In case of chromic acid burns, dilute sodium thiosulphate is generally used. In case of an exposure, going out for a fresh air and consulting a doctor is advised.
It may also be oxidized easily as it is a strong oxidizing agent. It may cause fire or explosion by contacting with organic substances. Therefore, it is advised to be cautious when mixing it with flammable substances. Although it is not flammable on his own, it must be kept off from direct fire or high temperatures. It may also lose stability in high temperatures. It must never be provided to city network because it is highly hazardous for environment.