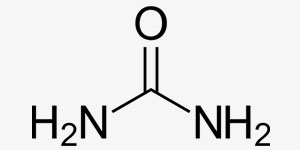
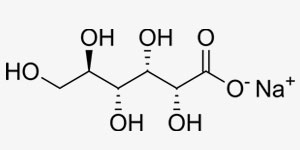
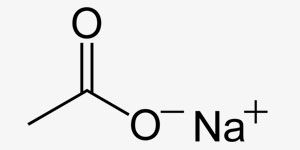
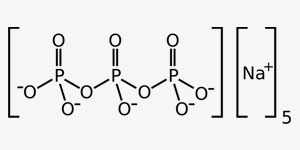
Salts are ionic compounds that contain anyons and cations. Their electrical charge is 0. When they are dissolved in water, they may yield alcali or neutral solvents and they are classified as such. They are dissolved by ionisation in water and they are good conductors for electricity.
Salts can be acquired via alcali reactions, acid-metal reactions or ion-metal reactions. They have a wide array of uses. NaCl, or table salt, is the most known salt.
Salts have a very large chemical family. While, some salts are completely natural and harmless, some are hazardous for humans.